- Depolarizing blockade
SCh, the only depolarizing NMBD, is composed of two ACh molecules linked together via an acetyl moiety. SCh binds to the α-subunits of the nicotinic AChR leading to depolarization of the postjunctional membrane. Because SCh is not degraded by AChE as rapidly as ACh, it persistently depolarizes the motor endplate, leading to inactivation of the voltage-gated sodium channels in the perijunctional zone that are necessary for propagation of the depolarization. Induction doses of SCh produce a rapid onset (about 1 minute) of a transient agonist effect (eg, muscle twitch) followed by skeletal muscle paralysis lasting 4 to 6 minutes. These characteristics make SCh a common choice for facilitating rapid tracheal intubation.
- SCh’s neuromuscular-blocking effect terminates when the drug diffuses away from the AChRs and is rapidly hydrolyzed by plasma cholinesterase (produced in the liver and also referred to as pseudocholinesterase) to succinylmonocholine and then, more slowly, to succinic acid and choline. This enzyme is not the same as AChE and is not found in the synaptic cleft. However, inhibitors of AChE affect both enzymes to different degrees.
- Side effects of SCh are related to its agonist effects at both the nicotinic and muscarinic AChRs. These include the following:
- Myalgias, especially in the muscles of the abdomen, back, and neck. It is attributed to muscle fasciculations and observed more frequently in females and younger patients after minor surgical procedures.
- Cardiac dysrhythmias. SCh has no direct effect on the myocardium. However, ganglionic stimulation may increase heart rate and blood pressure in adults. Alternatively, SCh may stimulate muscarinic receptors at the sinus node, producing sinus bradycardia, a junctional rhythm, or even asystole, particularly in children and following repeated exposure within a short time interval (eg, 5 minutes) in adults. Pretreatment of children with intravenous (IV) atropine immediately before SCh reduces the occurrence of bradyarrhythmias.
- Elevated serum potassium. SCh-mediated depolarization may raise serum potassium by 0.5 to 1.0 mEq/L by exaggerating the usual transmembrane ionic flux. However, life-threatening hyperkalemia and cardiovascular collapse may occur in patients with major burns, massive tissue injuries, extensive denervation of skeletal muscle, or upper motor neuron diseases. This effect is attributed to a proliferation of extrajunctional AChRs or damaged muscle membranes and a massive release of potassium upon stimulation. In patients with burns, the period of greatest risk is from 2 weeks to 6 months after the burn has been sustained. However, it is recommended to avoid SCh after the first 24 hours and for 2 years from the time of the injury. Patients with mild elevations of potassium related to renal failure may usually safely receive SCh.
- Increased intraocular pressure, by 5 to 10 mm Hg, which transiently occurs 2 to 4 minutes following SCh administration. The mechanism of this rise is not clear. The use of SCh in open eye injuries is still acceptable for rapid sequence inductions (Chapter 26).
- Increased intragastric pressure (IGP) results from fasciculations of abdominal muscles. The risk for aspiration is related to the balance between this pressure and the lower esophageal sphincter (LES) pressure. There have been limited human studies that demonstrate an increase in LES tone that is greater than the increase in IGP. There has not been clinical evidence of an increased aspiration risk with use of SCh for RSI.
- Increase in intracranial pressure, but the magnitude and clinical importance of this are unclear (Chapter 25). There have not been reports of cerebral herniation with use of SCh.
- A history of malignant hyperthermia (MH) is an absolute contraindication to the use of SCh. Some degree of masseter muscle spasm may be a normal response to SCh, but severe jaw rigidity increases the risk that a fulminant MH episode may follow. Generalized muscle rigidity, tachycardia, tachypnea, and profound hyperpyrexia after SCh should alert the clinician to MH (Chapter 19).
- Pretreatment with a subparalyzing dose of a nondepolarizing NMBD (eg, cisatracurium 1 mg IV or rocuronium 3 mg IV) 2 to 4 minutes before SCh may blunt visible fasciculations but is not uniformly effective in attenuating the abovementioned side effects. Moreover, awake patients pretreated with a nondepolarizing NMBD may experience diplopia, weakness, or dyspnea. When pretreating for a rapid sequence induction, the subsequent IV dose of SCh should be increased to 1.5 mg/kg.
- Phase I blockade.Neuromuscular blockade (NMB) produced by SCh can be separated into two phases. Phase I blockade (Figure 14.2) is the usual response to SCh as previously described and is characterized by the following:
- Transient muscle fasciculations followed by relaxation
Figure 14-2 Schematic representation of train-of-four (TOF) responses to a depolarizing (A) and a nondepolarizing (B) muscle relaxant, showing the control response before the relaxant (a) and afterward (b, c, d).
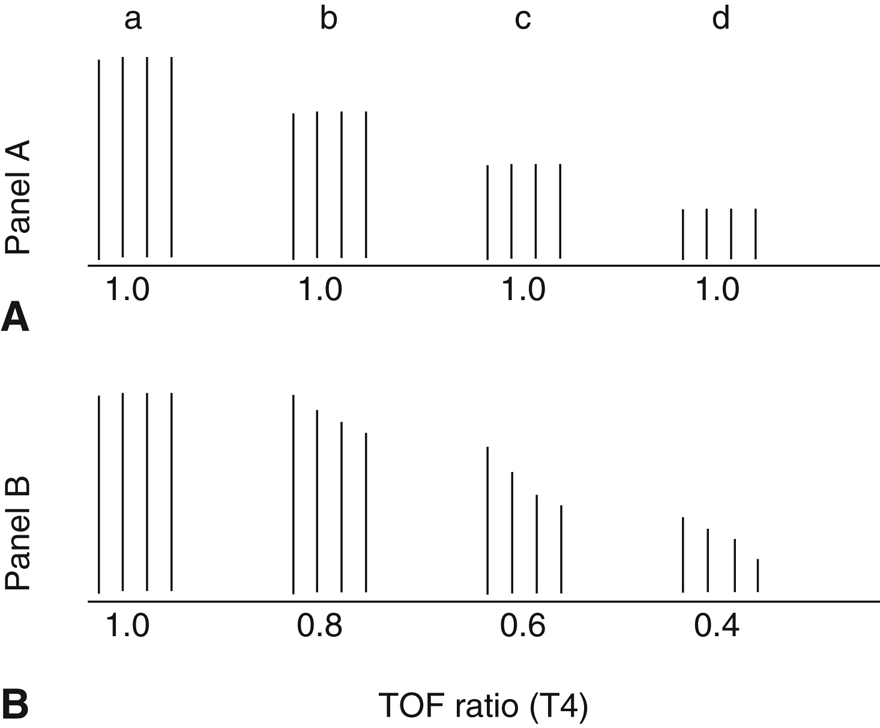
Note no fade with the depolarizing relaxant and progressive fade with the nondepolarizing relaxant.
- Absence of fade to tetanic or TOF stimulation (Section IV.C)
- Absence of post-tetanic potentiation (PTP; Section IV.C)
- AChE inhibitors potentiate rather than reverse the block
- Transient muscle fasciculations followed by relaxation
- Phase II blockade is most likely to occur after repeated or continuous administration of SCh when the total dose exceeds 3 to 5 mg/kg. Phase II blockade is thought to be secondary to repeated channel opening, causing distortion of the normal electrolyte balance and desensitizing the junctional membrane to further depolarization. It has some of the characteristics of a nondepolarizing blockade:
- Fade after tetanic or TOF stimulation (Section IV.C)
- Presence of PTP (Section IV.C)
- Tachyphylaxis (increasing dose requirement to achieve same depth of NMB)
- Prolonged recovery
- Partial or complete reversal by AChE inhibitors
- Prolonged blockade following SCh may be caused by low levels of plasma cholinesterase, a drug-induced inhibition of plasma cholinesterase activity, or a genetically atypical enzyme.
- Decreased plasma cholinesterase levels are found in severe liver or kidney disease, starvation, carcinomas, hypothyroidism, burn patients, decompensated cardiac failure, the last trimester of pregnancy and several days postpartum, and after therapeutic radiation.
- Inhibition of plasma cholinesterase occurs with the use of organophosphorus compounds (eg, echothiophate eye drops and insecticides), AChE inhibitors (eg, neostigmine, pyridostigmine, and donepezil), chemotherapeutic agents (eg, cyclophosphamide and nitrogen mustard), oral contraceptives, glucocorticoids, esmolol, and monoamine oxidase inhibitors. Plasma cholinesterase levels are not usually altered by hemodialysis.
- Several genetic variants of plasma cholinesterase exist: normal (N), atypical (A), fluoride resistant (F), and silent (S). Homozygous atypical cholinesterase (A–A, prevalence 0.04%) results in prolonged (2-3 hours) skeletal muscle paralysis and respiratory insufficiency following a conventional dose of SCh. Heterozygous atypical cholinesterase (N–A, prevalence 4%) results in only modest prolongation of effect.
- The dibucaine number is a laboratory assay used to characterize plasma cholinesterase abnormality. Normally, the local anesthetic dibucaine inhibits plasma cholinesterase activity by about 80% (dibucaine number of 80), whereas A–A plasma cholinesterase is inhibited by about 20% (dibucaine number of 20). In N–A, dibucaine numbers range from 30 to 65. The comparable fluoride number ranges from 0 to 60. N–F individuals (prevalence 0.005%) have slight prolongation of SCh effect, a normal dibucaine number, and a reduced fluoride number. A heterozygous silent N–S individual (prevalence 0.005%) has slightly prolonged effect, but dibucaine and fluoride numbers are normal. Homozygotes F–F and S–S are extremely rare.
- Patients exhibiting prolonged blocks after a single administration of SCh require sedation and continued intubation until there is a return of twitches. The NMB at this point is similar to that seen with nondepolarizing NMBD. Once fade with TOF is observed, reversal with neostigmine/glycopyrrolate is possible. Blood assays should be performed to determine total cholinesterase and, dibucaine number, and fluoride numbers.
- Nondepolarizing blockade is produced by reversible competitive antagonism of ACh at the α-subunits of the AChRs.
- It is characterized by the following (Figures 14.2 and 14.3):
- Absence of fasciculations
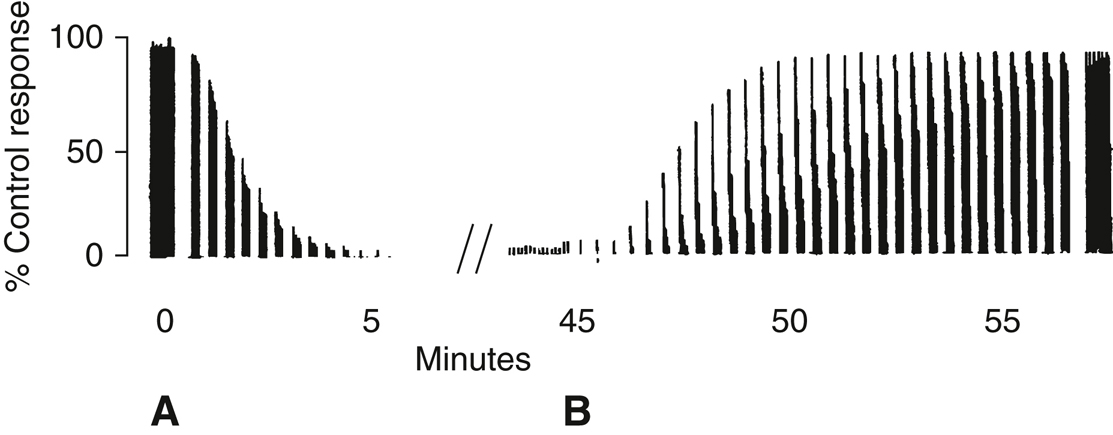
Figure 14-3
A, Electromyographic response to repeated train-of-four (TOF) stimulation after injection of a nondepolarizing agent. Each vertical bar is composed of four individual twitch responses. Fade of TOF response eventually leaves only one twitch (approximately 90% blockade).
B, Reversal of the blockade by neostigmine and atropine given 45 minutes later shows a progressive recovery of the TOF response and reduction in fade with a TOF ratio of 0.9.
- Fade during tetanic and TOF stimulation (Section IV.C)
- Presence of PTP (Section IV.C)
- Antagonism of depolarizing blockade
- Potentiation by other nondepolarizing NMBDs and volatile anesthetic agents
- Reversal by AChE inhibitors
- Absence of fasciculations
- The clinical pharmacology of the commonly used nondepolarizing NMBDs is outlined in Table 14.1. Synergistic blockade may occur when aminosteroid NMBDs are administered together with benzylisoquinolines. When NMBDs with similar chemical class are combined, the effect is additive.
- Cisatracurium is a benzylisoquinoline (see short description of fellow benzylisoquinolines atracurium and mivacurium in Table 14.1) of intermediate duration. High molar potency results in a relatively slow onset time (see rocuronium section). The drug is cleared primarily by Hofmann elimination, and its duration of action is largely independent of renal or hepatic function; it is, however, influenced by body temperature and pH given reliance on plasma enzymatic activity. Specifically, hypothermia may prolong duration of action, and hyperthermia may shorten duration of action.
- Vecuronium is a lipophilic, aminosteroid NMBD that is readily absorbed by the liver and excreted into the bile. One of the metabolites, 3-desacetylvecuronium, has neuromuscular-blocking properties (about 50% to 70% of the potency of vecuronium) and is eliminated by the kidneys. Vecuronium has a prolonged clinical effect in elderly patients and those with liver disease and renal failure, as a result of reduced clearance and extended elimination half-life. Vecuronium has no significant effects on heart rate and blood pressure, but it inhibits histamine N-methyltransferase and might potentiate effects such as flushing and hypotension when histamine is released by drugs such as morphine and vancomycin.
- Rocuronium is an analogue of vecuronium that has lower potency. The large intubating dose results in a fast onset time because a larger number of molecules reach the NMJ per circulation time. At a dose of 0.6 mg/kg, maximal blockade can be achieved between 1.8 and 2.7 minutes in adults, per product packaging. Increasing the dose to 1.2 mg/kg (four times the ED95) shortens the time even more but significantly prolongs the duration of action, which can be highly variable among patients. This drug is often chosen when a rapid sequence induction is required but SCh is contraindicated. Clearance of rocuronium occurs as unchanged drug in bile and through renal excretion. Administration of the drug to patients with renal failure may result in a longer duration of action, especially after repeated doses or continuous infusion, though this effect is unpredictable and varied.
- The cardiovascular side effects of nondepolarizing NMBDs are summarized in Table 14.2
- It is characterized by the following (Figures 14.2 and 14.3):