Diagrammatic Representation of Osmosis Depicting Water Molecules (Open Circles) and Solute Molecules (Solid Circles) Separated by a Semipermeable Membrane Water Molecules Move Across the Semipermeable Membrane to the Area of Higher Concentration of Solute Molecules. Osmotic Pressure is the Pressure that Would Have to Be Applied to Prevent Continued Movement of Water Molecules.
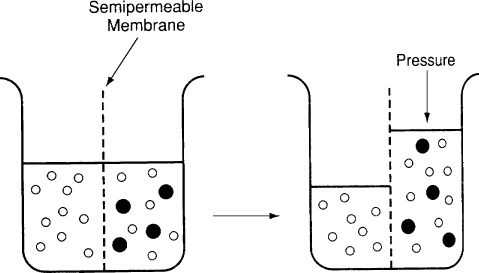
(From Ganong WF. Review of Medical Physiology. 21st ed. New York, NY: Lange Medical Books/McGraw-Hill; 2003.)
Diagrammatic representation of osmosis depicting water molecules (open circles) and solute molecules (solid circles) separated by a semipermeable membrane. Water molecules move across the semipermeable membrane to the area of higher concentration of solute molecules. Osmotic pressure is the pressure that would have to be applied to prevent continued movement of water molecules.