- PO (Adults and Children
 12 yr): Most infections — 200 mg every 12 hr; Skin and skin structure infections — 400 mg every 12 hr; Urinary tract infections/pharyngitis — 100 mg every 12 hr; Gonorrhea — 200 mg single dose.
12 yr): Most infections — 200 mg every 12 hr; Skin and skin structure infections — 400 mg every 12 hr; Urinary tract infections/pharyngitis — 100 mg every 12 hr; Gonorrhea — 200 mg single dose. - PO (Children 2 mo–12 yr): Pharyngitis/tonsillitis/otitis media/acute maxillary sinusitis — 5 mg/kg every 12 hr (not to exceed 200 mg/dose).
Renal Impairment
- PO (Adults): CCr <30 mL/min — ↑dosing interval to every 24 hr.

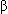 -lactamase-producing strains),
-lactamase-producing strains),