AUTHORS: Emily Sauck, DO, MBA and Anthony Sciscione, DO
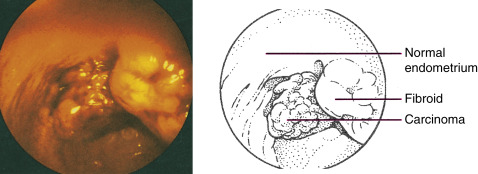
Endometrial cancer, also called endometrial carcinoma (EC), is cancer of the endometrium, which is the lining of the uterus. Traditionally, EC was divided into two types: Type 1, which are estrogen-driven, and type 2, which are not estrogen driven. However, EC is now more commonly subdivided into different types based on histology-how the cells appear under the microscope (Table 1).
TABLE 1 Pathogenetic Subsets of Endometrial Carcinoma
| Parameter | Type I | Type II |
|---|---|---|
| Age | 50s-60s | 60s-70s |
| Obesity | Common | Uncommon |
| Estrogenic stimuli | Common | Uncommon |
| Endometrium | Anovulatory | Atrophic |
| Precursor | Endometrial intraepithelial neoplasia | Presumed EmGD |
| Transition | Slow | Unknown |
| Type | Endometrioid | Papillary serous or mixed |
| Molecular genetics | MSI, PTEN mutation; loss of PAX2 | p53 mutation, 1p deletions; loss of PAX2 |
| Familial | Hereditary nonpolyposis colonic cancer syndrome | |
| Spread | Lymph nodes | Peritoneum |
| Concurrent ovarian | Common | Uncommon |
| Prognosis | Good | Poor |
EmGD, Endometrial glandular dysplasia; MSI, microsatellite instability.
From Crum CP et al: Diagnostic gynecologic and obstetric pathology, ed 3, Philadelphia, 2018, Elsevier.
Histologic types are summarized in Box 1 and include adenocarcinoma, uterine carcinosarcoma, squamous cell carcinoma, small cell carcinoma, transitional carcinoma, and serous carcinoma, with most endometrial cancers being adenocarcinomas and endometrioid cancer being the most common type of adenocarcinoma.
BOX 1 Endometrial Primary Adenocarcinomas
|
From Gershenson DM et al: Comprehensive gynecology, ed 8, Philadelphia, 2022, Elsevier.
| ||||||||||||||||
- In 2021, 66,570 new cases of uterine cancer are predicted in the U.S. The rate of new cases of EC was 28.1 per 100,000 based on 2014 to 2018 cases. Incidence was greater among white and black women compared with American Indian/Alaska Native, Hispanic, and Asian Pacific Islander women. It is the most common gynecologic malignancy in the U.S. and the most common type of cancer that affects the female reproductive organs.
- Unlike most cancers in the U.S., endometrial cancer is rising in both incidence and associated mortality.
Age: Most cases are diagnosed in postmenopausal women, with a median age of 63 yr.
Estrogen exposure/hormone imbalance: Whether from early menarche, late menopause, diabetes, nulliparity, tamoxifen use, polycystic ovary syndrome, or unopposed estrogen therapy, the more exposure the endometrium has to estrogen, the more a woman’s risk of developing EC increases.
- Obesity: Body mass index of 25 of greater is a major risk factor for EC.
- Genetics: Lynch syndrome increases the risk of EC (and ovarian, colon, and other types of cancers). Cowden syndrome: Relative risks can be found in Table 2.
TABLE 2 Risk Factors for Endometrial Cancer
| Factor | Relative Risk | ||
|---|---|---|---|
| Overweight (lbs): | |||
| 3.0 | |||
| 10.0 | |||
| Nulliparous: | |||
| 2.0 | |||
| 5.0 | |||
| Late menopause (>52 vs. 49 yr) | 2.4 | ||
| Diabetes mellitus | 2.7 | ||
| Unopposed estrogen therapy | 6.0 | ||
| Tamoxifen therapy | 2.0 | ||
| Sequential oral contraceptives | 7.0 | ||
| Combination oral contraceptives | 0.5 | ||
| Cowden syndrome (PTEN mutation) | Three- to fivefold increased risk | ||
| Hereditary nonpolyposis colonic cancer syndrome | 40%-60% lifetime risk | ||
| Family member with endometrial cancer | 3.4 | ||
From Crum CP et al: Diagnostic gynecologic and obstetric pathology, ed 3, Philadelphia, 2018, Elsevier.
∗Previously termed adenoacanthoma or adenosquamous carcinoma.