AUTHORS: Courtney Pfeuti, MD and Anthony Sciscione, DO
Molar pregnancy (hydatidiform mole) is a premalignant gestational disorder and is included in the spectrum of disorders characterized as gestational trophoblastic disease. Molar pregnancies are classified as complete or partial based on morphologic and pathologic examination. Both complete and partial molar pregnancies have an abnormal placenta with enlargement and swelling of the chorionic villi and hyperplasia of the villous trophoblastic cells. Most molar pregnancies are complete and are characterized by generalized hydropic villous changes with no fetal tissue. Partial moles are characterized by a mixture of large hydropic villi and normal placental tissue and often have fetal tissue present. Gestational trophoblastic disease (GTD) is composed of a spectrum of neoplastic conditions derived from the placenta. Whereas hydatidiform moles, gestational choriocarcinoma, and placental site trophoblastic tumor (PSTT) are histologic diagnoses, postmolar gestational trophoblastic neoplasia (GTN) is defined by clinical and laboratory criteria. The disease entities included in GTD have a wide variation in behavior, but GTN specifically refers to those with the potential for tissue invasion and metastases.
The risk of malignant sequelae (GTN) for a complete mole is 7% to 30% and for a partial mole is less than 2.5% to 7.5%.
| ||||||||||||||||
- 80% to 90% present with vaginal bleeding at 6 to 16 wk gestational age
- 28% with uterine enlargement greater than expected for gestational age
- 8% with hyperemesis gravidarum
- 1% with pregnancy-induced hypertension in the first or second trimester
- 9% to 25% with bilateral theca lutein cysts
- 15% will have a beta human chorionic growth hormone (beta hCG) >100,000 mIU/ml
- <10% with anemia
- 90% present with an incomplete or missed abortion
- 75% present with vaginal bleeding
- <10% will have a beta hCG of >100,000 mIU/ml
Table E1 Classic Presenting Signs and Symptoms of Complete and Partial Hydatidiform Moles
| Sign or Symptom | Complete Mole (%) (n = 306) | Partial Mole (%) (n = 81) |
|---|---|---|
| Vaginal bleeding | 97 | 73 |
| Excessive uterine size | 51 | 4 |
| Theca lutein cysts >6 cm | 50 | 0 |
| Preeclampsia | 27 | 3 |
| Hyperemesis | 26 | 0 |
| Hyperthyroidism | 7 | 0 |
| Trophoblastic emboli | 2 | 0 |
From Niederhuber JE: Abeloff’s clinical oncology, ed 6, Philadelphia, 2020, Elsevier.
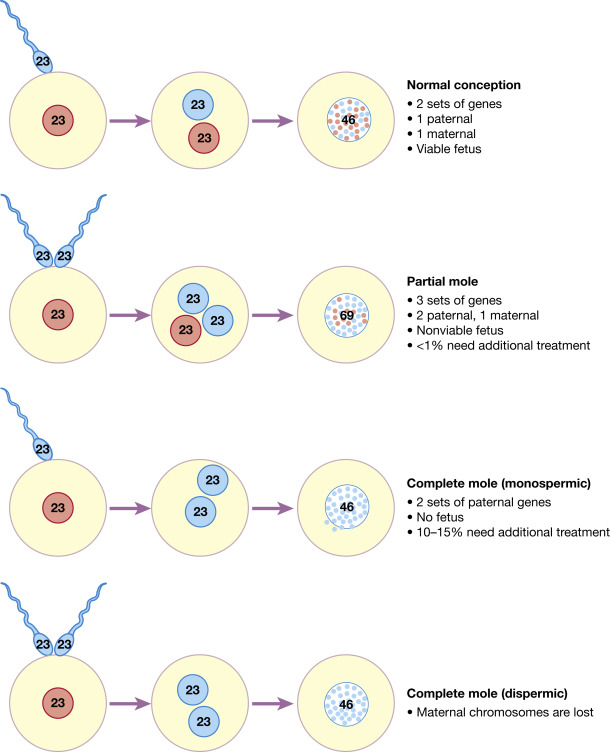
- Fertilization of an oocyte with absent or inactive maternal chromosomes (Fig. E1) and duplication of paternal chromosomes (85% to 90% are 46,XX) or, less commonly, fertilization of an empty oocyte with two sperm (46,XY)
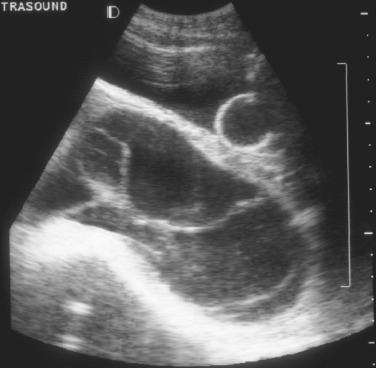
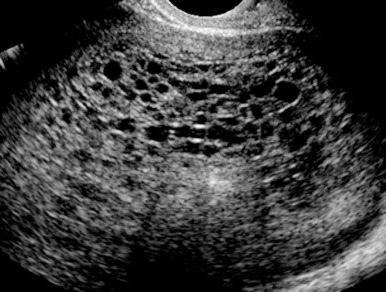
- Diffuse villous edema and enlargement and trophoblastic proliferation (Fig. E2) with no development of a fetus
Figure E1 Genetic makeup of normal pregnancy and partial and complete molar pregnancies.
From Magowan BA: Clinical obstetrics & gynecology, ed 4, New York, 2019, Elsevier.