Chronic Rx
- Box 1 summarizes the key recommendations for the treatment of blood cholesterol to reduce ASCVD risk in adults.
- The guidelines identify four high-risk groups that benefit from statin therapy:
- Patients with clinical ASCVD (Table 2, Fig. 1)
- LDL ≥190 mg/dl
- DM aged 40 to 75 yr and LDL 70 to 189 mg/dl
- 10-yr risk for ASCVD ≥7.5% and LDL 70 to 189 mg/dl
- The 10-yr risk of ASCVD is calculated with the risk calculator available at http://my.americanheart.org/cvriskcalculator.
- ASCVD events are reduced by using the maximum tolerated statin intensity in the aforementioned groups shown to benefit the most (Tables 3 and 4).
- Additional factors such as C-reactive protein >2 mg/L, primary LDL >160, genetic hyperlipidemias, family history of premature coronary heart disease (CHD), ankle-brachial index <0.9, and coronary artery calcium score (CAD) assessed with computed tomography may be used in patients who are not in one of four statin benefit groups and for whom a decision to initiate statin therapy is otherwise unclear. Statins are generally beneficial in patients at intermediate risk and selected patients with borderline risk who have a calcium score that is 100 or higher or who are in the 75th percentile or higher for their age, sex, and race. Statins should also be considered in persons with scores of 1 to 99, particularly if they are age 55 yr or older.
- Percent reduction in LDL cholesterol is used as a guide to compliance and adherence to therapy in the 2018 AHA/ACC Revised Clinical Practice Guidelines (Fig. 2). Studies have shown that compared with less-intensive LDL-C lowering, more intensive lowering reduces all-cause mortality and cardiovascular mortality; patients with higher baseline LDL-C have greater benefit.
- Moderate-intensity statin therapy should be continued for individuals >75 yr of age for secondary prevention. However, factors such as comorbidities, safety, and priorities of care should be considered before initiating statins for primary prevention of ASCVD.
- Adherence to lifestyle and to statin therapy should be reiterated with patients before the addition of a nonstatin drug.
- High-risk patients with a suboptimal response to statins who are unable to tolerate a recommended intensity or who are completely statin intolerant may benefit from the addition of a nonstatin cholesterol-lowering agent such as ezetimibe and/or Protein Convertase Subtilisin/Kexin 9 (PCSKS 9) inhibitor to reduce risk for major cardiovascular events.2 Combination therapies for LDL are summarized in Table 5.
- Ezetimibe inhibits cholesterol absorption in the intestine, whereas statins inhibit cholesterol production primarily in the liver. Ezetimibe is not as effective as most statins, but will reduce LDL by 15% to 22%. It can reduce the risk of heart attacks and strokes when taken alongside a statin, but there is little evidence it can do this if used on its own.
- PCSK9 binds to LDL receptors on hepatocytes, promotes receptor degradation, and prevents LDL-C clearance from the circulation thereby increasing serum concentrations of LDL-C. PCSK9 monoclonal antibody inhibitors alirocumab (Praluent), evolocumab (Repatha), and inclisiran (Leqvio), a PCSK9-directed small interfering RNA, are currently indicated as adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease, who require additional lowering of LDL cholesterol. PCSK9 inhibitors lower risk for ischemic cardiovascular events in persons with stable CAD and elevated atherogenic lipoproteins despite statin therapy. These medications are administered by subcutaneous injection and are expensive.
- The management of metabolic syndrome includes weight reduction, increased physical activity, and treatment of hypertension, elevated triglycerides, and low HDL cholesterol.
- According to recent studies, each 40 mg/dl reduction in LDL cholesterol by statin therapy confers a 20% reduction in ASCVD. In other words, a relative risk reduction of 30% in ASCVD by moderate-intensity therapy and 45% by high-intensity therapy has been approximated.
- Recent trials have shown that bempedoic acid, an inhibitor of ATP citrate lyase, reduces LDL cholesterol. The addition of bempedoic acid to maximally tolerated statin therapy did not lead to a higher incidence of overall adverse events than placebo and led to significant lowering of LDL cholesterol.
- Recent trials have shown that bempedoic acid, an inhibitor of ATP citrate lyase, reduces LDL cholesterol. The addition of bempedoic acid to maximally tolerated statin therapy did not lead to a higher incidence of overall adverse events than placebo and led to significant lowering of LDL cholesterol.
- Table 6 summarizes oral drugs affecting lipoprotein metabolism.
TABLE 6 Drugs Affecting Lipoprotein Metabolism
| Drug Class | Agents and Daily Doses | Lipid/Lipoprotein Effects | Side Effects | Contraindications |
|---|
| HMG-CoA reductase inhibitors (statins) | Lovastatin (10-40 mg)
Pravastatin (10-80 mg)
Simvastatin† (5-80 mg)
Fluvastatin (20-40 mg)
Atorvastatin (10-80 mg)
Rosuvastatin (5-40 mg)
Pitavastatin (2-4 mg) | LDL↓ 20%-60%
HDL↑ 5%-15%
TG↓ 7%-30% | Myalgias, myositis
Increased liver enzymes
New-onset diabetes (with intensive therapy)
Unproven concerns about memory loss | Active or chronic liver disease
Pregnancy
Concomitant use of certain drugs∗ |
| Bile acid sequestrants | Colestipol (5-20 g)
Colesevelam‡ (2.6-3.8 g)
Cholestyramine (4-16 g) | LDL↓ 15%-30%
HDL↑ 3%-5%
TG No change or increase | Gastrointestinal distress, constipation, drug interaction, hypertriglyceridemia
Decreased absorption of fat-soluble vitamins | TG >300 mg/dl
GI motility disorder |
| Omega-3 fatty acids | Fish oils (4-6 g) | TG↓ 45%
HDL↑ 13% | Increased bleeding time
Nausea | Caution with anticoagulant therapy |
| Nicotinic acid | Immediate release (niacin) (1.5-3 g)
Extended release (Niaspan) (1-2 g) | LDL↓ 5%-25%
HDL↑ 15%-35%
TG↓ 20%-50% | Flushing
Hyperglycemia
Hyperuricemia (or gout)
Upper GI distress
Hepatotoxicity | Chronic liver disease
Severe gout
Diabetes
Peptic ulcer disease
Pregnancy/lactation |
| Fibric acids | Gemfibrozil (600 mg bid)
Fenofibrate (45-145 mg) | LDL↓ 5%-20%
HDL↑ 10%-20%
TG↓ 20%-50% | Dyspepsia
Gallstones
Myopathy (especially with concomitant use of gemfibrozil and statins) | Severe renal disease (dose adjustment for fenofibrate)
Severe hepatic disease
Caution with statins
Can worsen LDL cholesterol |
| Ezetimibe (cholesterol absorption inhibitor) | Ezetimibe (10 mg) | LDL↓ 18%
HDL↑ 1%
TG↓ 8% | Abdominal pain; myalgias | Liver disease
Avoid with resins and fibrates |
GI, Gastrointestinal; HDL, high-density lipoprotein; HMG-CoA, 3-hydroxy-3-methylglutaryl coenzyme A; LDL, low-density lipoprotein; TG, triglyceride.
Modified from The National Cholesterol Education Program, JAMA 285:2486, 2001. In Boyden TF et al: Implementing new guidelines in the management of blood cholesterol, Am J Med 127:705, 2014.
TABLE 5 Combination Therapies for LDL
| Statin plus ezetimibe | Ezetimibe added to a statin may further reduce LDL by 20% or more and reduce triglycerides by 7%-13%.
The combination provides equivalent LDL reduction to a fourfold increase in statin dose.
Daily ezetimibe added to a low-dose statin given 2-3 times/wk can improve tolerance. Combination pills containing statin and ezetimibe are available.
Most common side effects reflect those of the individual drugs.
Combination ezetimibe and simvastatin has been shown to decrease cardiovascular events in patients with renal disease and acute coronary syndrome. |
| Statin and PCSK9 inhibitors | This combination is the most effective known treatment for hypercholesterolemia.
There are no known negative interactions between these two therapies.
This combination reduces CHD events more than statin alone. |
| Statin plus bile acid sequestrants | Bile acid sequestrants in combination with statins further decrease LDL from 24% to 60%.Cholestyramine and colestipol can interfere with the absorption of statins. Colesevelam does not affect statin absorption.
The statin-colesevelam combination is not ideal for patients with high triglycerides but may be useful in those with type 2 diabetes mellitus because colesevelam reduces glycemia. |
| Statin plus niacin | Adding niacin to a statin can lower LDL by 10% to 20%, in addition to beneficial effects on triglycerides. When used in combination with a statin, the maximum dose of niacin should be 2000 mg/day.
This combination in subjects with already low LDL levels did not reduce CHD events. |
| Bile acid sequestrants plus niacin | Before the availability of statins, bile acid sequestrants plus niacin were used to lower LDL in high-risk patients.
The availability of colesevelam and extended-release niacin has made this combination tolerable for many patients who are unable to use statins. |
| Ezetimibe plus bile acid sequestrants | Ezetimibe inhibits cholesterol absorption, and sequestrants enhance cholesterol excretion through conversion to bile acids. The combination can have additive effects.
This combination is useful for patients who cannot take statins. |
CHD, Coronary heart disease; LDL, low-density lipoprotein; PCSK9, proprotein convertase subtilisin/kexin type 9.
From Melmed S et al: Williams textbook of endocrinology, ed 14, St Louis, 2019, Elsevier.
Figure 2 A and B, Summary of 2018 ACC/AHA Cholesterol Guideline Recommendations for Statin and Nonstatin TherapyBackground Color Denotes Class and Level of Evidence: Green = Class I (Strong) Recommendation; Yellow = Class Iia (Reasonable) Recommendation; Blue = Grade A (High) Level of Evidence; Orange = Grade B (Moderate) Level of Evidence. Ascvd, Atherosclerotic Cardiovascular Disease; Cac, Coronary Artery Calcium; LDL-C, Low-Density Lipoprotein Cholesterol.
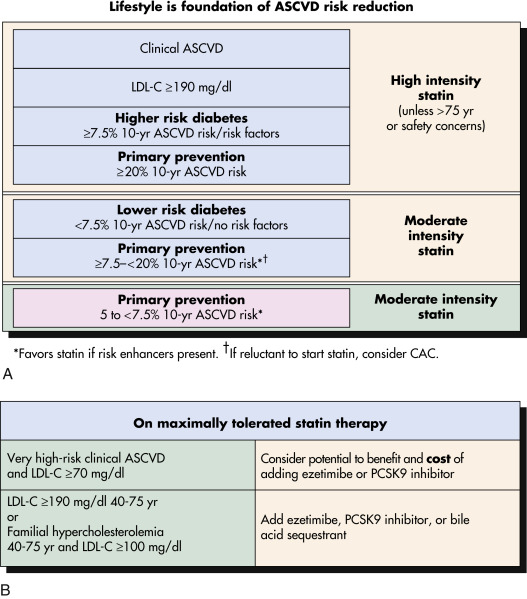
From Goldman L, Shafer AI: Goldman’s Cecil medicine, ed 26, St Louis, 2019, Elsevier.
TABLE 3 Statin Benefit Groups and Recommended Therapy
| Statin Benefit Group | High Intensity | Moderate Intensity | Additional Testing |
|---|
| Clinical ASCVD | Yes | Consider† | None |
| Primary LDL-C >190 mg/dl | Yes | Consider† | None |
| Diabetes without ASCVD and 10-yr risk ≥7.5%∗ | Yes | Consider† | None |
| Diabetes without ASCVD and 10-yr risk <7.5%∗ | Consider‡ | Yes | Case-by-case |
| Primary prevention and 10-yr risk ≥7.5%∗ | Consider‡ | Yes | Case-by-case |
| Primary prevention and 10-yr risk <7.5%∗ | Consider‡ | Consider‡ | Case-by-case |
ASCVD, Atherosclerotic cardiovascular disease; LDL-C, low-density lipoprotein cholesterol.
From Boyden TF et al: Implementing new guidelines in the management of blood cholesterol, Am J Med 127:705, 2014.
TABLE 4 High-, Moderate-, and Low-Intensity Statin Therapy∗
| Statin Therapy | Daily Dose |
|---|
High Intensity†
↓LDL-C ≥50% | Moderate Intensity‡
↓LDL-C 30<50% | Low Intensity§
↓LDL-C <30% |
|---|
| Atorvastatin | (40||)-80 mg | 10(20)mg | |
| Rosuvastatin | 20(40)mg | (5)10 mg | |
| Simvastatin | | 20-40 mg¶ | 10 mg |
| Pravastatin | | 40(80)mg | 10-20 mg |
| Lovastatin | | 40 mg | 20 mg |
| Fluvastatin | | 80 mg (Fluvastatin XL) | 20-40 mg |
| Fluvastatin | | 40 mg∗∗ | |
| Pitavastatin | | 2-4 mg | 1 mg |
FDA, U.S. Food and Drug Administration; LDL-C, low-density lipoprotein cholesterol; XL, extended-release.
Reprinted with permission of the authors: Stone NJ et al: 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines, J Am Coll Cardiol 63(25, Part B):2889-2934, 2014.
Figure 1 Initiating statin therapy in individuals with clinical ASCVD.
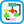
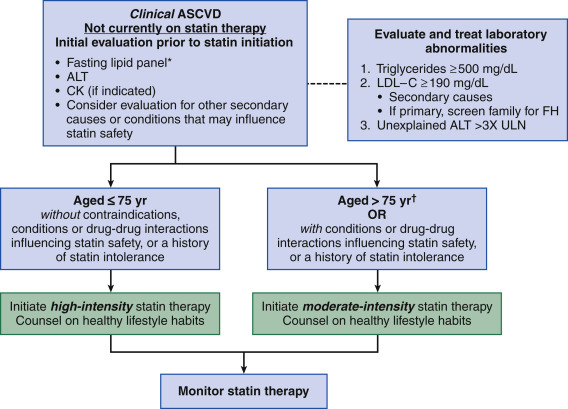
∗Fasting lipid panel is preferred. In a nonfasting individual, a nonfasting non-HDL-C >220 mg/dl may indicate genetic hypercholesterolemia that requires further evaluation or a secondary etiology. If nonfasting triglycerides are >500 mg/dl, a fasting lipid panel is required.†It is reasonable to evaluate the potential for ASCVD benefits and for adverse effects and to consider patient preferences in initiating or continuing a moderate- or high-intensity statin in individuals with ASCVD >75 yr of age. ALT, Alanine transaminase; ASCVD, atherosclerotic cardiovascular disease; CK, creatine kinase; FH, familial hypercholesterolemia; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; ULN, upper limit of normal.
Modified from Stone NJ et al: 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines, J Am Coll Cardiol, 2013. In Mann DL et al: Braunwald’s heart disease, ed 10, Philadelphia, 2015, Elsevier.
TABLE 2 Atherosclerotic Cardiovascular Disease
- Coronary heart disease: Acute coronary syndromes, history of myocardial infarction, stable or unstable angina, coronary or other arterial revascularization
- Stroke or transient ischemic attack
- Peripheral arterial disease
|
BOX 1 2013 ACC/AHA Summary of Key Recommendations for the Treatment of Blood Cholesterol to Reduce ASCVD Risk in Adults
- Heart-healthy lifestyle habits should be encouraged for all individuals
- The appropriate intensity of statin therapy should be initiated or continued
- Clinical ASCVD∗
- Age 75 yr or less and no safety concerns: High-intensity statin (class I, level A)
- Age 75 yr or safety concerns: Moderate-intensity statin (class I, level A)∗
- Primary prevention: Primary LDL-C 190 mg/dl or greater
- Rule out secondary causes of hyperlipidemia (class I, level B)
- Age 21 yr or older: High-intensity statin (class I, level B)
- Achieve at least a 50% reduction in LDL-C (class IIa, level B)
- LDL-C lowering nonstatin therapy may be considered to further reduce LDL-C (class IIb, level C)
- Primary prevention: Diabetes, 40 to 75 yr of age, and LDL-C 70 to 189 mg/dl
- Moderate-intensity statin (class I, level A)
- Consider high-intensity statin when 7.5% or greater 10-yr ASCVD risk using the Pooled Cohort Equations (class IIa, level B)‡
- Primary prevention: No diabetes, 40 to 75 yr of age, and LDL-C 70 to 189 mg/dl
- Estimate 10-yr ASCVD risk using the Risk Calculator based on the Pooled Cohort Equations in those not receiving a statin; estimate risk every 4 to 6 yr (class I, level B)
- To determine whether to initiate a statin, engage in a clinician-patient discussion of the potential for ASCVD risk reduction, adverse effects, drug-drug interactions, and patient preferences
- Reemphasize heart-healthy lifestyle habits and address other risk factors (class IIa, level C)
- 7.5% or greater 10-yr ASCVD risk: Moderate- or high-intensity statin (class I, level A)
- 5% to 7.5% 10-yr ASCVD risk: Consider moderate-intensity statin (class IIa, level B)
- Other factors may be considered: LDL-C 160 mg/dl or greater, family history of premature ASCVD, hs-CRP 2.0 mg/L or greater, CAC score 300 Agatston units or greater, ABI less than 0.9, or lifetime ASCVD risk (class IIb, level C)
- Primary prevention when LDL-C is less than 190 mg/dl and age is less than 40 or more than 75 yr, or less than 5% 10-yr ASCVD risk
- Statin therapy may be considered in selected individuals (class IIb, level C)
- Statin therapy is not routinely recommended for individuals with NYHA class II-IV heart failure or who are receiving maintenance hemodialysis
- Regularly monitor adherence to lifestyle and drug therapy with lipid and safety assessments
- Assess adherence, response to therapy, and adverse effects within 4 to 12 wk following statin initiation or change in therapy (class I, level A)
- Measure a fasting lipid panel (class I, level A)
- Do not routinely monitor ALT or CK unless symptomatic (class IIa, level C)
- Screen and treat type 2 diabetes according to current practice guidelines. Heart-healthy lifestyle habits should be encouraged to prevent progression to diabetes (class I, level B)
- Anticipated therapeutic response: Approximately 50% or greater reduction in LDL-C from baseline for high-intensity statin and 30% to 50% for moderate-intensity statin (class IIa, level B)
- Insufficient evidence for LDL-C or non-HDL-C treatment targets from RCTs
- For those with unknown baseline LDL-C, an LDL-C less than 100 mg/dl was observed in RCTs of high-intensity statin therapy
- Less than anticipated therapeutic response:
- Reinforce improved adherence to lifestyle and drug therapy (class I, level A)
- Evaluate for secondary causes of hyperlipidemia if indicated (class I, level A)
- Increase statin intensity, or if on maximally tolerated statin intensity, consider addition of nonstatin therapy in selected high-risk individuals (class IIb, level C)§
- Regularly monitor adherence to lifestyle and drug therapy every 3 to 12 mo once adherence has been established. continue assessment of adherence for optimal ASCVD risk reduction and safety (class I, level A)
- In individuals intolerant of the recommended intensity of statin therapy, use the maximally tolerated intensity of statin (class I, level B). If there are muscle or other symptoms, establish that they are related to the statin (class IIa, level B)
|
ABI, Ankle brachial index; ACC, American College of Cardiology; AHA, American Heart Association; ALT, alanine transaminase; ASCVD, atherosclerotic cardiovascular disease; CAC, coronary artery calcium; CK, creatine kinase; hs-CRP, high sensitivity C-reactive protein; LDL-C, low-density lipoprotein cholesterol; MI, myocardial infarction; NYHA, New York Heart Association; RCTs, randomized clinical trials; TIA, transient ischemic attack.From 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines, J Am Coll Cardiol 63(25 Pt B):2889-2934, 2014.


