Central Nervous System Effects
Mental impairment is not detectable in volunteers breathing 1,600 ppm (0.16%) nitrous oxide (unlikely that impairment of mental function in the personnel who work in the operating room using modern anesthetic scavenging techniques can result from inhaling trace concentrations of anesthetics).
Cerebral metabolic oxygen requirements are decreased in parallel with drug-induced decreases in cerebral activity.
Drug-induced increases in cerebral blood flow (CBF) may increase intracranial pressure in patients with space-occupying lesions.
Desflurane and isoflurane are similar in terms of increases in CBF and the preservation of reactivity to carbon dioxide (Fig. 4-10).
Anesthetic-induced increases in CBF occur within minutes of initiating administration of the inhaled drug and whether blood pressure is unchanged or decreased, emphasizing the cerebral vasodilating effects of these drugs.
Seizure Activity. Enflurane (not desflurane or sevoflurane) can produce fast frequency and high voltage on the electroencephalograph that often progresses to spike-wave activity that is indistinguishable from changes that accompany a seizure.
Evoked Potentials. Volatile anesthetics cause dose-related decreases in the amplitude and increases in the latency of the cortical component of median nerve somatosensory evoked potentials, visual evoked potentials, and auditory evoked potentials.
Intracranial Pressure (ICP). Inhaled anesthetics produce increases in ICP that parallel increases in CBF produced by these drugs. Patients with space-occupying intracranial lesions are most vulnerable to these drug-induced increases in ICP.
Circulatory Effects. Inhaled anesthetics produce dose-dependent and drug-specific circulatory effects. The circulatory effects of desflurane and sevoflurane parallel many of the characteristics of older inhaled anesthetics with desflurane most closely resembling isoflurane, whereas sevoflurane has characteristics of both isoflurane and halothane.
Mean Arterial Pressure (Figs. 4-11 and 4-12)
Heart Rate (Fig. 4-13). A small dose of opioid (morphine in the preoperative medication or fentanyl intravenously immediately before induction of anesthesia) can prevent the heart rate increase associated with isoflurane and presumably the other volatile anesthetics.
Cardiac Output and Stroke Volume (Fig. 4-14)
Systemic Vascular Resistance. Isoflurane, desflurane, and sevoflurane, but not halothane, decrease systemic vascular resistance when administered to healthy human volunteers (Fig. 4-15).
Duration of Administration. Administration of a volatile anesthetic for 5 hours or longer is accompanied by recovery from the cardiovascular depressant effects of these drugs (compared with measurements at 1 hour, the same MAC concentration after 5 hours is associated with a return of cardiac output toward predrug levels) (Figs. 4-16 and 4-17).
Cardiac Dysrhythmias. The ability of volatile anesthetics to decrease the dose of epinephrine necessary to evoke ventricular cardiac dysrhythmias is greatest with the alkane derivative halothane and minimal to nonexistent with the ether derivatives isoflurane, desflurane, and sevoflurane (Figs. 4-18, 4-19, 4-20).
Spontaneous Breathing. Circulatory effects produced by volatile anesthetics during spontaneous breathing are different from those observed during normocapnia and controlled ventilation of the lungs (reflects the impact of sympathetic nervous system stimulation due to accumulation of carbon dioxide [respiratory acidosis] and improved venous return during spontaneous breathing).
Coronary Blood Flow. Volatile anesthetics induce coronary vasodilation.
Preexisting Diseases and Drug Therapy
Volatile anesthetics decrease myocardial contractility of normal and failing cardiac muscle by similar amounts, but the significance is greater in diseased cardiac muscle because contractility is decreased even before administration of depressant anesthetics.
Valvular heart disease may influence the significance of anesthetic-induced circulatory effects (peripheral vasodilation produced by isoflurane and presumably also desflurane and sevoflurane is undesirable in patients with aortic stenosis but may be beneficial by providing afterload reduction in those with mitral or aortic regurgitation).
Prior drug therapy that alters sympathetic nervous system activity (antihypertensives,
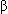 -adrenergic antagonists) may influence the magnitude of circulatory effects produced by volatile anesthetics.
-adrenergic antagonists) may influence the magnitude of circulatory effects produced by volatile anesthetics.
Ventilation Effects. Inhaled anesthetics produce dose-dependent and drug-specific effects on the (a) pattern of breathing, (b) ventilatory response to carbon dioxide, (c) ventilatory response to arterial hypoxemia, and (d) airway resistance. The Pao2 predictably declines during administration of inhaled anesthetics in the absence of supplemental oxygen.
Patterns of Breathing
Inhaled anesthetics, except for isoflurane, produce dose-dependent increases in the frequency of breathing (isoflurane increases the frequency of breathing similarly to other inhaled anesthetics up to a dose of 1 MAC but at a concentration of >1 MAC does not produce a further increase in the frequency of breathing).
Tidal volume is decreased in association with anesthetic-induced increases in the frequency of breathing.
The net effect of these changes is a rapid and shallow pattern of breathing during general anesthesia. The increase in frequency of breathing is insufficient to offset decreases in tidal volume, leading to decreases in minute ventilation and increases in Paco2.
Ventilatory Response to Carbon Dioxide. Volatile anesthetics produce dose-dependent depression of ventilation characterized by decreases in the ventilatory response to carbon dioxide and increases in the Paco2 (Fig. 4-21).
Surgical stimulation increases minute ventilation by about 40% because of increases in tidal volume and frequency of breathing. The Paco2, however, decreases only about 10% (4 to 6 mm Hg) despite the larger increase in minute ventilation (see Fig. 4-22).
The reason for this discrepancy is speculated to be an increased production of carbon dioxide resulting from activation of the sympathetic nervous system in response to painful surgical stimulation.
Increased production of carbon dioxide is presumed to offset the impact of increased minute ventilation on Paco2.
Management of Ventilatory Depression
The predictable ventilatory depressant effects of volatile anesthetics are most often managed by institution of mechanical (controlled) ventilation of the patient’s lungs (inherent ventilatory depressant effects of volatile anesthetics facilitate the initiation of controlled ventilation).
Assisted ventilation of the lungs is a questionably effective method for offsetting the ventilatory depressant effects of volatile anesthetics (apneic threshold [maximal Paco2 that does not initiate spontaneous breathing] is only 3 to 5 mm Hg lower than the Paco2 present during spontaneous breathing).
Ventilatory Response to Hypoxemia. All inhaled anesthetics, including nitrous oxide, profoundly depress the ventilatory response to hypoxemia that is normally mediated by the carotid bodies.
Airway Resistance and Irritability (Fig. 4-23)
Hepatic Effects
Hepatic blood flow during administration of desflurane and sevoflurane is maintained similar to isoflurane (Fig. 4-24). Maintenance of hepatic oxygen delivery relative to demand during exposure to anesthetics is uniquely important in view of the evidence that hepatocyte hypoxia is a significant mechanism in the multifactorial etiology of postoperative hepatic dysfunction.
Drug Clearance. Volatile anesthetics may interfere with clearance of drugs from the plasma as a result of decreases in hepatic blood flow or inhibition of drug-metabolizing enzymes.
Liver Function Tests. Transient increases in the plasma alanine aminotransferase activity follow administration of enflurane and desflurane, but not isoflurane administration, to human volunteers (Fig. 4-25).
Hepatotoxicity. Postoperative liver dysfunction has been associated with most volatile anesthetics, with halothane receiving the most attention (Fig. 4-26).
Renal Effects. Volatile anesthetics produce similar dose-related decreases in renal blood flow, glomerular filtration rate, and urine output (most likely reflect the effects of volatile anesthetics on systemic blood pressure and cardiac output). Preoperative hydration attenuates or abolishes many of the changes in renal function associated with volatile anesthetics.
Fluoride-induced nephrotoxicity. All volatile anesthetics introduced since methoxyflurane undergo significantly less metabolism, and their decreased solubility compared with methoxyflurane means that substantial amounts of the anesthetic are exhaled and thus are not available for hepatic metabolism to fluoride.
Vinyl Halide Nephrotoxicity. Carbon dioxide absorbents containing potassium and sodium hydroxide react with sevoflurane (degradation product produced in greatest amounts). Compound A is a dose-dependent nephrotoxin in rats. The amount of compound A produced under clinical conditions has consistently been far below those concentrations associated with nephrotoxicity in animals.
Skeletal Muscle Effects
Neuromuscular Junction. Volatile anesthetics produce dose-dependent enhancement of the effects of neuromuscular-blocking drugs, with the effects of enflurane, isoflurane, desflurane, and sevoflurane being similar and greater than halothane.
Malignant Hyperthermia. All volatile anesthetics including desflurane and sevoflurane can trigger malignant hyperthermia in genetically susceptible patients even in the absence of concomitant administration of succinylcholine.
Obstetric Effects. Volatile anesthetics produce similar and dose-dependent decreases in uterine smooth muscle contractility and blood (Fig. 4-27).
Resistance to Infection. Many normal functions of the immune system are depressed after patient exposure to the combination of anesthesia (likely the result of surgical trauma and the subsequent endocrine and inflammatory responses).
Peripheral Neuropathy. Humans who chronically inhale nitrous oxide for nonmedical purposes may develop a neuropathy characterized by sensorimotor polyneuropathy that is often combined with signs of posterior lateral spinal cord degeneration resembling pernicious anemia (ability of nitrous oxide to oxidize irreversibly the cobalt atom of vitamin B12 such that activity of vitamin B12-dependent enzymes is decreased).